Anatomy & Physiology
Nervous system
The nervous system consists of a main nerve ring, which encircles the oesophagus inside the Aristotle’s lantern (Nichols 1967; Ruppert et al. 2004). Radial nerves run from this, along the underside of the test. The spines, tube feet and pedicellariae have a sensory role in the nervous system, and are lined with sensory cells to accommodate this role (Ruppert et al. 2004). Many sea urchins are negatively phototactic, which means they respond to light and search for shade, either in crevices or by covering themselves with objects collected by their tube feet (Ruppert et al. 2004). T. alexandri displays this behaviour.
Water Vascular System (WVS)
The WVS is characteristic of the phylum Echinodermata (Ruppert et al. 2004). It is a fluid-filled system consisting of a series of canals connecting on the inside of the body (Nichols 1971). Seawater is the main component of the WVS, as well as coelomocytes, which make up the main cellular component of the immune system in echinoderms (Pinsino et al. 2007). Its main function is related to the control of the tube feet. It does this by generating and distributing the necessary pressure for their operation (Nichols 1971). The WVS is also involved in gas exchange and waste excretion (Nichols 1971). There are three main parts that make up the WVS: the ring canal, stone canal and peripheral system (Nichols 1971). Together, the stone canal and ring canal are considered the central parts of the system. The ring canal encircles the oesophagus and the stone canal originates at the ring canal, leading to an exterior opening, the hydropore (Nichols 1971; Ruppert et al. 2004). The peripheral system also arises from the ring canal. It consists of tube feet and radial canals enclosed by the ambulacrum (Nichols1971). These radial canals are arranged pentaradially and maintain the fluid pressure, which is used along with ampullae, to control the tube feet (Nichols1971). To accomplish this, the radial canal walls are muscular and elastic (Ruppert et al. 2004). The central parts of the WVS supply each radial canal with coelomic fluid.
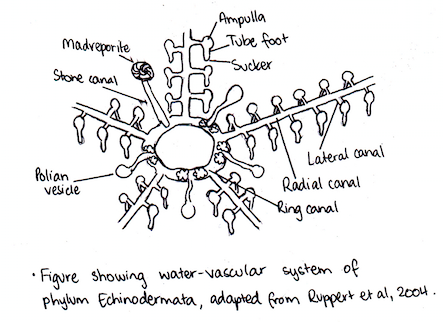
Internal Transport, Gas Exchange and Excretion (Information summarised from Ruppert et al. 2004)
Coelomic fluid is the main circulatory medium transported around the body of echinoids. They also have a well-developed hemal system and peristomial gills. In some echinoids, the peristomial gills are thought to be the gas exchange surface, whereas others use tube feet for this process. Coelomic fluid is pumped from the peristomial gills around the body by a combination of muscles and ossicles. Excretion occurs at the aboral surface, through the anus.
Adhesion
Living in the ocean, where conditions are variable, means animals have had to develop ways to resist wave action, desiccation and carry out their day-to-day activities. Adhesion is an essential part of life in the ocean. An adhesive bond is formed in echinoids when attaching, burrowing, feeding and in locomotion (Flammang 1995). This adhesion is the responsibility of tube feet (Flammang 1995), which are evident in many variations. There are two mechanisms in which tube feet can attach to substrate; they either mould the tube foot disc to the substrate or secrete an adhesive to fill space (Santos et al. 2005). The mechanism used to attach to substratum has been found to be dependent on the surface (Santos et al. 2005), with stronger adhesion on rough surfaces. For stronger adhesion, the first mechanism is used; the combination of moulding the tube foot disc and secreting a thin film of adhesive allows this stronger attachment. The epidermal adhesive areas of tube feet have four cell types: secretory, neurosecretory, sensory andsupport cells (Flammang 1995). T.alexandri posses disc-ending podia, which use both chemical (adhesive secretions) and mechanical (tube feet) attachment (Flammang 1995). The specialized glandular areas of the disc-ending podia, where adhesive secretions are released, are at the disk surface (Flammang 1995). These adhesive secretions are released by the secretory cells (Flammang 1995). As well as releasing adhesive secretions, echinoids also release de-adhesive secretions when detaching themselves from the substrate (Santos et al. 2005). These de-adhesive secretions are released by the neurosecretory cells (Flammang 1995). Testing the adhesive strength of species has been studied; this is done by measuring their tenacity. Tenacity is a measure of adhesive force per unit area and is measured in Pascals (Pa) (Flammang et al. 2005).
Tube feet (podia)
Tube feet are external extensions of the WVS and perform a variety of functions (Smith 1979), including chemo-sensory reception, adhesion, gas exchange, feeding and locomotion (Flammang et al. 2005; Leddy & Johnson 2000; Nichols 1971; Smith 1979). They have diversified into a range of structures (Flammang 1995) and can either perform a number of these functions or be specialised. Due to their variety of functions, they have been classified as the most advanced hydraulic organs in the animal kingdom (Flammang et al. 2005). Tube feet are broken into two types: principal tube feet and peristomial tube feet (Nichols 1961). Principal tube feet arise from pore-pairs of the test and are different from peristomial tube feet, which extend from the peristomial membrane (Nichols 1961). Peristomial tube feet function in tasting and end in a disc that is distinctly broader than the stem, but is lacking a sucker (Smith 1979). Internally, tube feet are connected to the ampulla (Leddy & Johnson 2000).
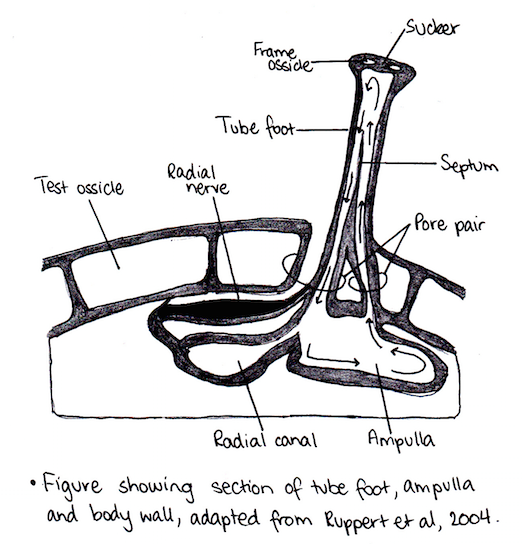
There are six categories of podia that have been formed based on morphology: disc-ending, pencillate, knob-ending, lamel-late, ramified and digitate (Flammang 1995). The podia characteristic of family Temnopleuridae are disc-ending. Disc-ending podia consist of a basal extensible cylinder, a stem and an apical extremity, which is enlarged and flattened forming the disc (Flammang 1995). The disc has a supportive skeleton enclosed in it and is made up of a combination of large (ossicles) and small (spicules) calcareous plates (Flammang 1995). They also have a circular plate of connective tissue at the distal end and a thick ring around the outside of the podium (Flammang 1995). Disc-ending podia, like all other podia, are controlled by the combination of muscles and the WVS; however, there are different muscle systems present between different classes of echinoids (Flammang 1995). Disc-ending podia possess a levator muscle, which is longitudinally arranged (Flammang 1995). It attaches distally to the centre of the diaphragm and is used to create suction when attaching to substrate (Flammang 1995). This is the main form of attachment for disc-ending podia. There is no differentiation of podia on the oral or aboral surface of urchins within the order Camarodonta (Fenner 1973); however, the podia situated around the mouth are thought to function in sensory perception. The suckered podia on the aboral surface aid in covering the body of the urchin with objects (Fenner1973). This can be for shade or camouflage. Tube feet on the oral surface have a thicker stem wall and wider disc; this feature allows for stronger adhesion(Santos & Flammang 2004). The strength of the tube feet is related to the strength of the adhesion (Santos & Flammang 2004). Due to this, the strength of the tube feet varies between species based on the environment they inhabit.
The proximal extensible cylinder (stem) connects the disc to the test (Santos & Flammang 2004). The stem wall of tube feet consist of four tissue layers (Nichols 1961; Santos & Flammang 2004); the inner myomesothelium surrounding the water-vascular lumen, connective tissue layer, nerve plexus and outer epidermis covered by cuticle. The connective tissue is embedded with spicules and the two epithelial layers are ciliated (Nichols 1961). The disk is formed by the expansion of the distal epithelium and connective tissue layer (Nichols 1961). Between the connective tissue layer and outer epidermis is a fine tract of nerve fibres (Smith 1979). A cylindrical sheath of collagenous connective tissue fibres run longitudinally up the stem except for the innermost fibres, which form a thin circular layer (Smith 1979). Lying internally from this connective tissue is a thin layer of longitudinal retractor muscle fibres (Smith 1979). The epithelial layer of tube feet is thick and includes mucus glands, which secrete adhesive material. At the distal end of tube feet are a disc or sensory pad (Smith1979). A calcareous rosette can also be seen at the distal end of the foot and functions in support and strength (Nichols 1961; Smith 1979). There are two pores where the tube feet connect to the test and a septum running up the centre of the podium (Fenner 1973).
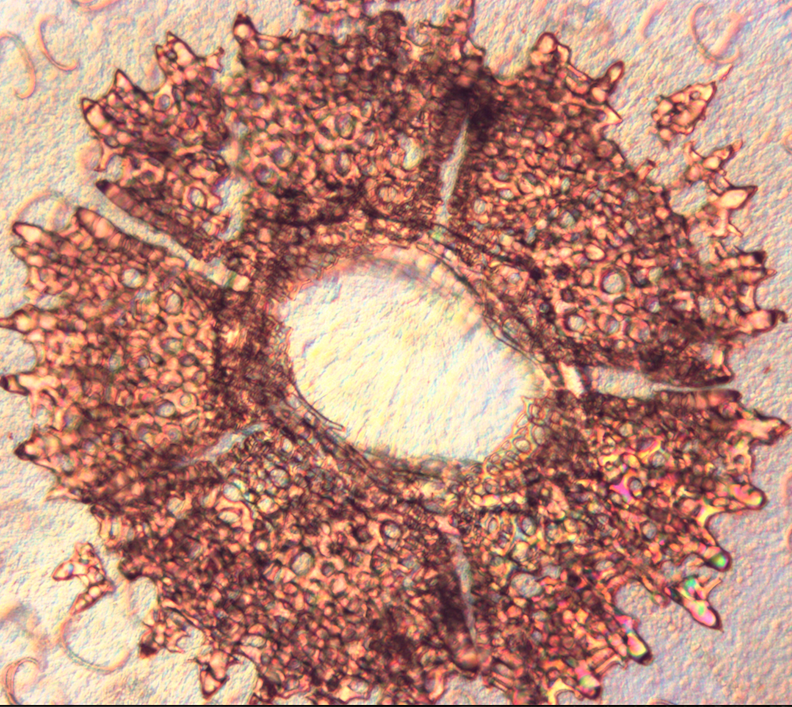 |
Distal view of Temnopleurus alexandri tube foot
Photo: Monique Parisi |
The tube feet exhibit three basic movements in mobility: protraction, flexion and retraction (Flammang 1995). Contraction and protraction of the tube feet is controlled by muscular action and hydrostatic pressure (Flammang 1995). The action of surrounding tube feet also assists protraction and contraction (Nichols 1971). To contract the tube feet, the outer epithelium is folded and the wall of the tube foot is thickened (Florey & Cahill 1977). To extend the tube feet, the longitudinal muscle contracts, forcing the lateral canal valve to close and pushing water into the foot (Ruppert et al.2004). Contraction is also necessary for the refilling of mucus glands (Nichols 1971).
As well as functioning in locomotion, echinoid tube feet have been shown to respond to light intensity (Millot 1954). A recent study by Luter et al. (2011) identified two important visual genes expressed in tube feet. These photoreceptor cells were found at the base of the tube feet and the rim of the tube foot disc. This could explain the phototactic behaviour displayed by urchins and in particular T. alexandri, which can be seen covering its oral surface with materials such as shells and seaweed. Podia that function in respiration have four characteristics that separate them from non-respiratory podia (Fenner 1973). They have a separation of the two ciliary currents within the podium, increased surface area and counter current across the surfaces of both podia and ampullae, and finally they are favourably positioned on the test for this respiratory function (Fenner 1973).
 |
Temnopleurus alexandri displaying covering behaviour
Photo: Monique Parisi |
Ampullae
Each radial canal has a number of lateral canals extending from it leading to a tube foot (Ruppert et al. 2004). At the end of each lateral canal is an ampulla, which is a small, muscular sac that controls the extension of the foot by allowing water to flow into the foot. Specifically, contraction of ampullae forces coelomic fluid into the podia, extending it (Leddy & Johnson 2000). Coelomic fluid is forced back through the ampulla when the longitudinal muscles of the tube feet contract (Leddy & Johnson 2000). Fenner (1973) and Nichols (1971) described Temnopleuridae ampullae as flattened, leaf-like sacs that connect through pore pairs to the tube feet by a small tube. Based on this, it can be expected that T. alexandri would have these ampullae as they belong to the Temnopleuridae family. Other ampullae have been described as cyclindrical or bulbous (Smith 1979). The ciliary currents within the amupllae are guided by septae (Fenner 1973). The way in which the ampullae function allow the tube feet to act independently of each other (Smith 1979). The ampullae are separated from the radial water vessel by a one way-valve and the wall of the ampulla consists of collagenous connective tissue with branching contractile fibres, which are arranged circularly (Smith 1979).
|
|
Video footage filmed in the lab of Temnopleurus alexandri using a combination of spines and
tube feet in locomotion
Footage: Monique Parisi
|
|